 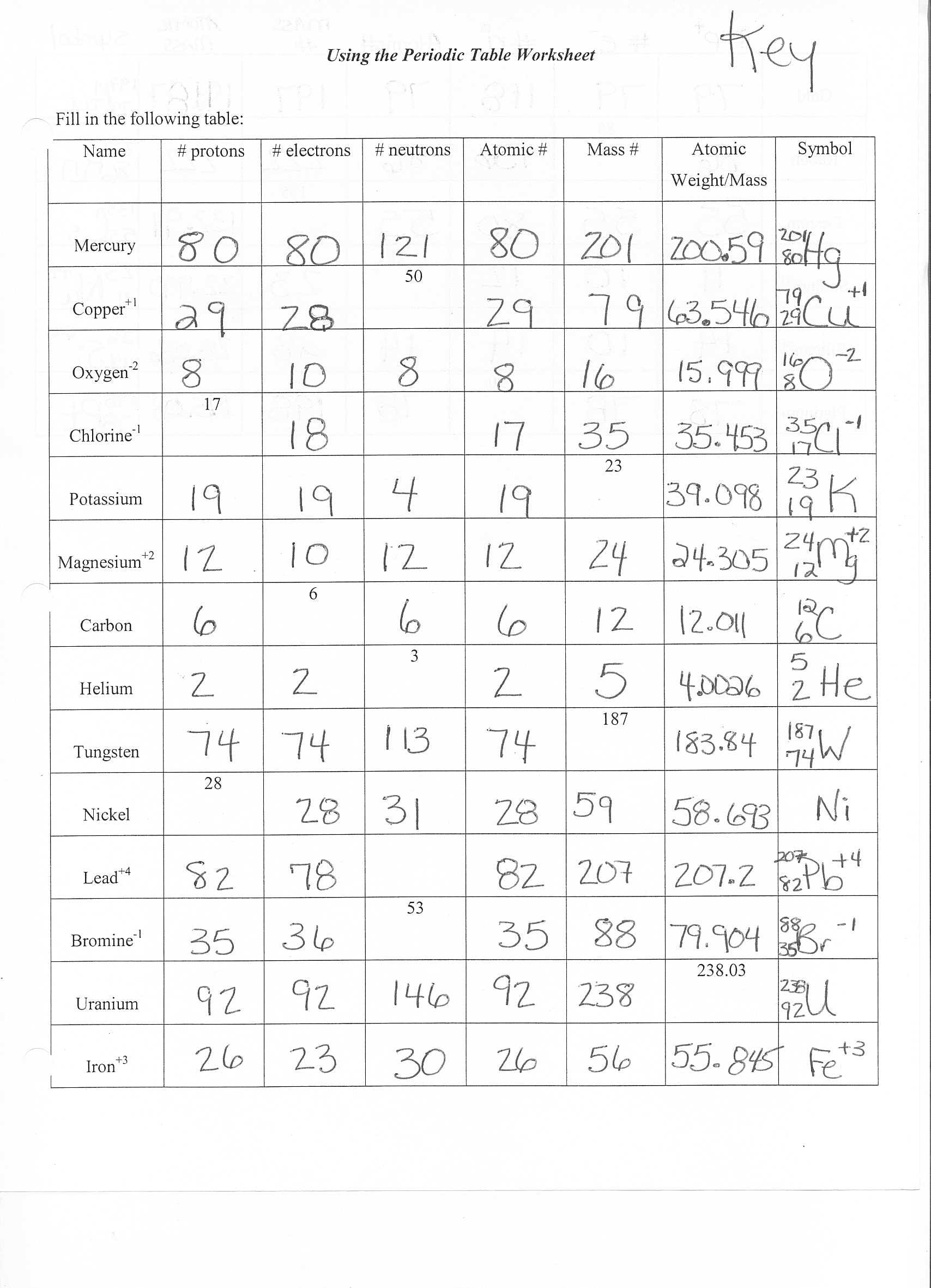
Position of electrons, protons and neutrons in atomġ3. Which of the following statements about the electron is incorrect?.The position and velocity of the electrons in the orbit cannot be determined simultaneouslyĪnswer: The position and velocity of the electrons in the orbit cannot be determined simultaneouslyĨ. What happens to the atomic number during a chemical reaction?ĩ. Atoms that have the same mass number and different atomic number are called?ġ0. Which of the following determines the chemical properties?ġ1. Total number of orbitals associated with third shell will be _.ġ2. Which of the following properties of atom could be explained correctly by Thomson Model of atom?.Electrons revolve in different orbits around the nucleus.The electron in the orbit nearest the nucleus has the lowest energy.Energy of the electrons in the orbit is quantised.Atomic Structure Multiple Choice Questions and Answersġ. The orientation of atomic orbitals depends on theirģ. The isotopes of a neutral atom of an element differ in which of these?Ĥ. The maximum mass of an atom is concentrated in which of these?ĥ. An atom differs from an ion with respect to which of the following?Ħ. The electronic configuration of atomic number 20 of an atom is which of the following?ħ. Which of the following statements does not form a part of Bohrs model of hydrogen atom? We have provided these questions in PDF format which you can Download by clicking the link as “Atomic Structure Multiple Choice Questions And Answers Pdf” provide below. These MCQs are very helpful in your preparation for the National Eligibility cum Entrance Test-UG (NEET) and JEE Mains. So these Class 11 Chemistry Chapter 2 Important Questions With Answers cover all the topics of Structure of Atom. Preparation of these questions has been taken from various books including NCERT, Previous year’s question papers, and model papers. These questions are based on the chapter on Structure of Atom. This MCQ on Structure Of Atom Class 11 Questions And Answers Pdf is prepared by our LiveMCQs team for NEET.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
